我室王斌举教授在 ACS Catalysis 上发表论文:Emergence of Function from Nonheme Diiron Oxygenases: A Quantum Mechanical/Molecular Mechanical Study of Oxygen Activation and Organophosphonate Catabolism Mechanisms by PhnZ
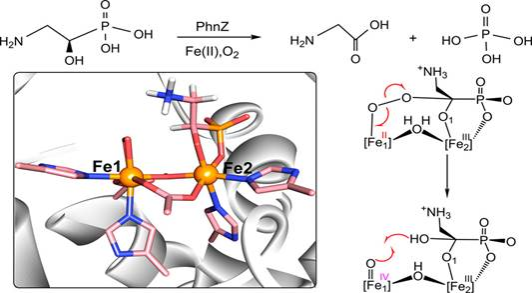
摘要:Organophosphonate (Pn) catabolism constitutes the major source of inorganic phosphate (Pi), which is essential for the synthesis of genetic and cellular components. Unlike various C–P hydrolases, the nonheme diiron enzyme PhnZ catalyzes the oxidative transformation of Pn (R)-2-amino-1-hydroxyethylphosphonic ((R)-OH-AEP) into glycine and Pi. Full enzymatic molecular mechanisms, starting from the initial oxygen activation to the C–P bond activation, are not yet understood. Using quantum mechanics/molecular mechanics (QM/MM) calculations, we uncover the oxygen activation and organophosphonate catabolism mechanisms by PhnZ. In contrast to previous studies, our calculations show that the Fe1(II)-catalyzed homolytic cleavage of an O–O bond of a Fe2(III)Fe1(II)-hemiketal intermediate is involved in the catalysis, affording the Fe1(IV) = O species that is responsible for the O–H bond activation of the geminal diolate intermediate. The following C–P cleavage and O–P formation were found to be facile in the resulting O-centered substrate radical species. On the basis of QM/MM calculations, we propose a catalytic cycle of PhnZ that is in line with the experimental kinetics. In particular, our study demonstrates that the catalytic cycle of PhnZ involves the spin-regulated electron transfer, Fe(II)-catalyzed homolytic cleavage of the O–O bond, general acid–base catalysis of a bridging OH ligand, and the cooperative catalysis of Fe1 and Fe2. Such wonderful catalysis of PhnZ would expand our understanding of the catalysis and function of nonheme diiron oxygenases.
文章链接:https://pubs.acs.org/doi/10.1021/acscatal.1c05116