我室吴川六教授在 J. Am. Chem. Soc. 上发表论文:Design and Ribosomal Incorporation of Noncanonical Disulfide-Directing Motifs for the Development of Multicyclic Peptide Libraries
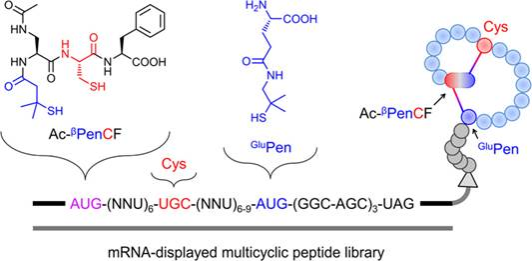
摘要:The engineering of naturally occurring disulfide-rich peptides (DRPs) has been significantly hampered by the difficulty of manipulating disulfide pairing. New DRPs that take advantage of fold-directing motifs and noncanonical thiol-bearing amino acids are easy-to-fold with expected disulfide connectivities, representing a new class of scaffolds for the development of peptide ligands and therapeutics. However, the limited diversity of the scaffolds and particularly the use of noncanonical amino acids [e.g., penicillamine (Pen)] that are difficult to be translated by ribosomes greatly hamper the further development and application of these DRPs. Here, we designed and synthesized noncanonical bisthiol motifs bearing sterically obstructed thiol groups analogous to the Pen thiol to direct the folding of peptides into specific bicyclic and tricyclic structures. These bisthiol motifs can be ribosomally incorporated into peptides through a commercially available PURE system integrated with genetic code reprograming, which enables, for the first time, the in vitro expression of bicyclic peptides with two noncanonical and orthogonal disulfide bonds. We further constructed a bicyclic peptide library encoded by mRNA, with which new bicyclic peptide ligands with nanomolar affinity to proteins were successfully selected. Therefore, this study provides a new, general, and robust method for discovering de novo DRPs with new structures and functions not derived from natural peptides, which would greatly benefit the field of peptide drug discovery.
文章链接:https://pubs.acs.org/doi/10.1021/jacs.2c00216